SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

responding covid-19 pandemic
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic
What Wikipedia / Google Says
About ‘Coronavirus Covid-19 Pandemic‘ ? :
Portraying All Stories
ON/ABOUT/FROM
‘Coronavirus Covid-19 Pandemic’
Add Story/ Your Say Until the Cause is Concluded
[*Select Category/Tag:
Coronavirus Covid-19 Pandemic
at Your Next Publish Screen.]

Anti-vaccine figure partners with Roku after YouTube banned him for sharing dangerous coronavirus misinformation

Broadcast and streaming platform Roku has added an online show from anti-vaccine activist Del Bigtree, who has repeatedly encouraged people to intentionally contract COVID-19. The program is listed as “educational” by Roku. In late July, YouTube removed Bigtree’s The HighWire channel for violating the platform’s policies after Media Matters reported on Bigtree using YouTube to spread dangerous medical misinformation.
During The HighWire’s latest broadcast, which aired on August 27 on Roku and also on his other biggest streaming platform, Facebook, he again argued that people should seek to contract COVID-19 in order to build natural herd immunity, a course of action that would lead to millions of deaths in the U.S.
Bigtree, who has no medical credentials, is a leading figure in the anti-vaccination movement through his anti-vaccine nonprofit organization Informed Consent Action Network and as the host of The HighWire, which is broadcast on Thursdays. A 2019 profile of Bigtree in the online parenting magazine Fatherly labeled him “dangerous” and said he “may be the most connected node in the anti-vaccine activist network.”
On August 19, The HighWire’s Twitter account teased the show’s “brand new ROKU Channel.” In an August 27 email to supporters, Bigtree wrote that following the YouTube ban, “we have started establishing relationships with companies who share in our vision of providing a new space for you to get real news, real science, transparent, and uncensored” and as part of that effort “we’ve also launched The HighWire on Roku!” Roku prohibits channel owners from publishing content that “place[s] individuals or groups in imminent harm” or material found to “contain false, irrelevant or misleading information.”
Bigtree’s Roku channel offers episodes dating back to July 30. During the July 30 broadcast, which was largely about his channel being banned from YouTube, Bigtree defended his claim that people should “catch this cold” by intentionally contracting COVID-19 “to create herd immunity” and said it was “incredible” that he was able to continue to broadcast live on Facebook.
In contrast to YouTube’s outright ban of Bigtree’s channel, Facebook — where Bigtree has over 348,000 followers — has taken a more piecemeal approach to containing his promotion of conspiracy theories and dangerous medical misinformation. Following Media Matters’ reporting, Facebook removed some videos where Bigtree made dangerous claims, but not others that contained similar dangerous claims. Facebook applied a fact-checking label to his August 27 broadcast — to note that Bigtree’s suggestion that the novel coronavirus was manmade is a conspiracy theory — but took no action regarding the dangerous claims he made during the broadcast that people should intentionally contract COVID-19 to build natural herd immunity.
During that broadcast, which is available on Bigtree’s new Roku channel, Bigtree argued that Americans should be willing to assume the same level of risk of dying of COVID-19 as we would if “China was attacking our borders.” Along those lines, Bigtree said, “I assure you, we would risk the lives of millions.” Bigtree then argued that the maximum number of Americans who could die from COVID-19 was “just nearing 200,000 total casualties, people who most likely were probably going to die this year anyway.” In fact, more than 183,000 Americans already have died of COVID-19 (and the actual death toll, based on total excess deaths, is even higher) and there is no end in sight to hundreds of Americans dying of the disease each day.
as per our monitoring this Story originally appeared * : ) here → *
Anti-vaccine figure partners with Roku after YouTube banned him for sharing dangerous coronavirus misinformation
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

‘Mix-and-match’ coronavirus vaccines to be tested
Ms Keenan, who is originally from Enniskillen, said: “I feel so privileged to be the first person vaccinated against Covid-19. It’s the best early birthday present I could wish for because it means I can finally look forward to spending time with my family and friends in the new year after being on my own for most of the year.”
as per our monitoring this Story originally appeared * : ) here → *
‘Mix-and-match’ coronavirus vaccines to be tested
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

Covid-19 Updates: First U.S. Vaccines Are About to Be Shipped as Virus Ravages America
Last Updated
Dec. 13, 2020, 11:50 p.m. ET
Dec. 13, 2020, 11:50 p.m. ETWith Pfizer’s vaccine cleared for emergency use on Friday, states eagerly began their own preparations to receive the vaccine within days. Deaths are rising sharply in college towns.
This briefing has ended. Follow our latest coverage of the coronavirus pandemic.
Here’s what you need to know:
UPS and FedEx say plans to ship the vaccine are underway.
A C.D.C. panel has endorsed the Pfizer vaccine for people 16 and over.
With the first injections expected to be given as early as Monday, this is how states are preparing.
Charley Pride, country music star, dies from complications of Covid-19.
The largest health care provider in the New York region is frantically prepping for the arrival of the vaccine.
Deaths from the virus have risen sharply in college towns — but few have been college students.
Britain, Canada and the U.S. start their vaccination campaigns, and other news from around the world.
An outbreak at an Illinois veterans home has killed more than a quarter of residents.
 UPS said it would transport doses of the vaccine from storage sites in Michigan and Wisconsin to its air cargo hub in Louisville, Ky.Credit…John Sommers Ii/Reuters
UPS said it would transport doses of the vaccine from storage sites in Michigan and Wisconsin to its air cargo hub in Louisville, Ky.Credit…John Sommers Ii/ReutersUPS and FedEx, normally rivals, are working side by side to ship the coronavirus vaccine developed by Pfizer and BioNTech, the first of the vaccines to win U.S. government approval.
The two shipping companies said they had put plans they had been working on for months into action after the Food and Drug Administration gave the vaccine emergency authorization late Friday.
Delivery of the first vaccines comes as the virus continues to rage across America, with officials reporting more than 207,000 new cases on Saturday. That brought the total number to more than 16 million, by far the most in the world, less than a week after the country surpassed 15 million. More than 3,000 deaths were reported for the first time on Wednesday, and the country’s total is approaching 300,000.
At a news conference on Saturday, Gen. Gustave F. Perna, the chief operating officer of Operation Warp Speed, the federal effort to bring a vaccine to market, said that boxes were being packed at Pfizer’s plant in Kalamazoo, Mich., and would be shipped to UPS and FedEx distribution hubs, where they would be dispersed to 636 locations across the country. Pfizer said shipping would start early Sunday morning.
Mr. Perna specified that 145 sites would receive the vaccine on Monday, 425 on Tuesday and 66 on Wednesday.
“Make no mistake, distribution has begun,” he said. UPS said it expected to start transporting the vaccine on Sunday morning, when employees stationed at Pfizer’s facility in Michigan will affix special Bluetooth- and radio-enabled tracking tags to each shipment. An aircraft waiting nearby will take the vaccine to the company’s Worldport air cargo hub in Louisville, Ky., a sprawling 5.2-million-square-foot sorting facility. Future shipments of the vaccine will also be transported by truck to the Louisville hub, which is up to a six-hour drive from Michigan.
“This is the moment we’ve been waiting for,” Wes Wheeler, president of the company’s health care division, said in an interview on Saturday. “We’ve been planning for months with daily calls, drilling down to really quite minute details.”
Mr. Wheeler said he planned to oversee the first vaccine shipments from a newly established 24/7 command center in Louisville, which will be staffed at any given time by teams of five to 10 people who will monitor each vaccine package as it moves through the UPS network.
Once the vaccine shipments arrive at the Louisville hub, they will be sorted alongside other packages and distributed to hospitals and other medical facilities. Every truck driver or airplane pilot will know if they are carrying a vaccine package, Mr. Wheeler said. Both UPS and FedEx have said that doses will arrive at their destinations a day after leaving the Pfizer facilities.
Even before the vaccine was approved, UPS had already started shipping out kits with the medical supplies needed to administer it, such as alcohol wipes and syringes, Mr. Wheeler told a Senate subcommittee this week. UPS and FedEx will split distribution of the vaccine throughout the country. After those shipments arrive, all Pfizer dosing sites will receive another shipment from UPS of 40 pounds of extra dry ice to keep the vaccines at a frigid temperature, he said.
“You have two fierce rivals here, and competitors, in FedEx and UPS, who literally are teaming up to get this delivered,” Richard Smith, a FedEx executive, told the Senate’s Subcommittee on Transportation and Safety on Thursday.
Both companies said the shipments would be closely tracked and monitored, and would be given priority over other packages. To ship its vaccine, Pfizer designed specialized containers packed with enough dry ice to keep a minimum of 975 doses cool for up to 10 days. Each comes with a tracking device.
Like UPS, FedEx said it would also affix its own tracking tags to vaccine shipments. Each UPS truck carrying the doses will have a device that tracks its location, temperature, light exposure and motion, Mr. Wheeler told the senators. The company’s trucks will have escorts, too, he said on Thursday. It is not clear whether he meant the local police or other government officials, or possibly private guards, and he declined to elaborate on that and other details in the interview, citing security concerns. But the trucks leaving Pfizer’s facility will be tracked “by the minute,” he said.
 Students at the Mast Academy in Miami arriving for classes in October. The vaccine has been approved for use in 16- and 17-year-olds.Credit…Lynne Sladky/Associated Press
Students at the Mast Academy in Miami arriving for classes in October. The vaccine has been approved for use in 16- and 17-year-olds.Credit…Lynne Sladky/Associated PressAn independent committee of experts advising the Centers for Disease Control and Prevention on Saturday afternoon recommended the Pfizer coronavirus vaccine for people 16 and older. That endorsement, which now only awaits final approval by Dr. Robert Redfield, the director of the C.D.C., is a key signal to hospitals and individual health care providers that they should proceed to inoculate patients.
It follows Friday night’s emergency use authorization of the vaccine by the Food and Drug Administration, which oversees licensing of medical products.
“Based on everything we saw from looking at the data, we’re very comfortable that the safety profile that was observed in 17- and 16-year-olds was acceptable,” Dr. Peter Marks, the F.D.A.’s top vaccine regulator, said at a news conference on Saturday.
The C.D.C. advisory committee, which typically meets three times a year to review amendments to routine schedules for child, adolescent and adult vaccines, has been engaged in numerous marathon-length sessions this fall to discuss a plethora of issues surrounding the introduction of limited supplies during a pandemic of the novel vaccine.
In meetings on Friday and Saturday, the panel’s heated discussion centered mainly on three areas: whether to recommend the vaccine for patients 16 and 17 years old, for pregnant and lactating women, and for patients who have had an anaphylactic reaction to other vaccines.
C.D.C. officials and scientists will review the debate and post more precise guidance about those specific groups and others on Sunday and throughout next week, as more information about the vaccine becomes known.
 Vail Health Hospital placed mock coronavirus vaccines into the hospital’s ultracold freezer on Tuesday in Colorado.Credit…Helen H. Richardson/The Denver Post, via Associated Press
Vail Health Hospital placed mock coronavirus vaccines into the hospital’s ultracold freezer on Tuesday in Colorado.Credit…Helen H. Richardson/The Denver Post, via Associated PressThis weekend, 2.9 million doses of the Pfizer and BioNTech vaccine are to begin traveling by plane and guarded truck from facilities in Michigan and Wisconsin to designated locations, mostly hospitals, in all 50 states.
The first injections are expected to be given by Monday to high-risk health care workers, the initial step toward the goal of inoculating enough Americans by spring to finally halt the spread of a virus that has killed nearly 300,000, sickened millions and upended the country’s economy, education system and daily life. There are now more than 16 million virus cases reported in the United States, according to a New York Times database.
The rapid development of the vaccine, and the F.D.A.’s emergency authorization of it on Friday night based on data showing it to be 95 percent effective, has been a triumph of medical science, but much in this complicated next stage could go wrong.
States say they have only a fraction of the funding they need from the federal government for staffing to administer the shot, for tracking who has received both doses of the vaccine — a booster is needed three weeks after the initial injection — and for other crucial pieces of the effort.
But for all the planning that has been done and contingencies that have been put in place in recent months, there is still a good deal of confusion. States are receiving initial allocations according to a federal formula based strictly on their adult population, but many hospitals say they still don’t know exactly how much they will get or when shipments will arrive. Some hospital systems are reeling from the news that their initial allocations will be much smaller than they had hoped.
One reason for the shortfall in initial supply is that federal officials decided to send out fewer than half of the 6.4 million doses they had planned for the first wave.
Although there is some variation among their plans, states are largely planning to follow recommendations from the Centers for Disease Control and Prevention about who gets vaccinated first: health care workers at high risk of exposure to the virus and residents of nursing homes and other long-term-care facilities, a population that has died from the virus at disproportionately high rates.
On Thursday, as an F.D.A. advisory committee debated whether to recommend authorization of the Pfizer vaccine, the first packages of supplies to administer it — vaccination record cards, masks, visors, information sheets and syringes — arrived at UPMC Presbyterian, a hospital in Pittsburgh.
 Charley Pride performing in New York in 1975.Credit…Bettmann Archive, via Getty Images
Charley Pride performing in New York in 1975.Credit…Bettmann Archive, via Getty ImagesCharley Pride, who was celebrated as a country music’s first Black superstar and known for hits such as “Kiss an Angel Good Mornin’” and “Is Anybody Goin’ to San Antone,” died on Saturday at age 86 while in hospice care in Dallas.
The cause of his death was complications of Covid-19, said Jeremy Westby, the singer’s publicist.
Lauded as a star who paved the way for other country music artists of color, Mr. Pride was praised for his contributions to the country music canon. In November, he received the Country Music Association’s Lifetime Achievement Award at the 54th annual Country Music Association Awards, where he also performed.
Some guests at the show, which was Mr. Pride’s last public event, were not wearing masks. However, event organizers said all protocols from the Centers for Disease Control and Prevention were followed.
Born March 18, 1934 in Sledge, Miss., Mr. Pride served in the army before moving to Montana to try to make it as a baseball player. There, he worked at a smelting plant and played semiprofessional baseball in East Helena, where he was paid $10 to sing the national anthem before games.
He began his country music career in the 1960s after signing with RCA Records, later quitting his factory job after his 1967 song “Just Between You and Me” became a Top 10 country hit.
As a Black man entering the country music industry in the 1960s, Mr. Pride’s career did not escape prejudice. Once radio stations learned he was Black, many refused to play his music, and during a 1970s award show, singer Loretta Lynn was told not to embrace him should he win an award she was presenting. (She did so anyway.)
“We’re not colorblind yet,” Mr. Pride wrote in his memoir, “Pride: The Charley Pride Story” (1994). “But we’ve advanced a few paces along the path, and I like to think I’ve contributed something to that process.”
During his career, Mr. Pride recorded over 50 hits that made it to Top 10 on the country charts, with over 20 hitting No. 1. Mr. Pride also racked up notable awards: Country Music Association’s male vocalist of the year in 1971 and 1972, Country Music Association’s entertainer of the year in 1971 and a lifetime achievement Grammy Award in 2017.
Dolly Parton said on Saturday she was “heartbroken” over Mr. Pride’s death, calling him one of her “dearest friends.”
“What a horrible, horrible virus,” she said on Twitter. “Charley, we will always love you.”
Mr. Pride is survived by his wife, Ebby Rozene Cohran Pride, and his children, Carlton, Charles and Angela.
 Freezers at Northwell Health at New Hyde Park, N.Y., awaiting doses of the coronavirus vaccine.Credit…Northwell Health
Freezers at Northwell Health at New Hyde Park, N.Y., awaiting doses of the coronavirus vaccine.Credit…Northwell HealthThe emergency authorization of the Pfizer-BioNTech vaccine on Friday night has set off a frenzy of preparations at hospitals and doctors’ offices across the United States. Among them is Northwell Health, the largest health care provider in the New York region, once the coronavirus epicenter in the United States.
Organizing the receipt and further distribution of the vaccine is a mammoth task. Northwell has set up nine vaccination centers equipped with ultracold freezers at or near its hospitals in Manhattan, Staten Island, Westchester County and Long Island, and plans to start vaccinating tens of thousands of employees as soon as the vaccine shipments arrive, probably on Monday, said Dr. Mark Jarrett, Northwell’s chief quality officer.
The health system, which has 56,000 employees who interact with patients, has already received shipments of kits with supplies for administering the vaccine, including syringes, alcohol swabs and gloves.
Northwell expects to have all of its workers — not just care providers like doctors and nurses, but also others such as cleaning personnel who work in intensive care units and patient rooms — vaccinated within five weeks with the Pfizer-BioNTech or the Moderna vaccines, Dr. Jarrett said.
Employees will be given appointments to come in for shots so they won’t have to stand in line and can maintain social distancing. A backup list of additional recipients will be drawn up in case anyone has to cancel an appointment at the last minute.
“The one thing we don’t want to do is ever waste vaccine,” Dr. Jarrett said. The Pfizer-BioNTech vaccine, which is transported on dry ice, has to be stored at minus 94 degrees Fahrenheit, and then prepared for use.
“Once you’ve defrosted and diluted it, you only have six hours to use it,” Dr. Jarrett said. “You want to certainly use every dose that’s in a vial.”
The health system is carefully scheduling employees for vaccination in various units and hospitals to prevent staffing problems from overburdening any single unit. The idea is to evenly spread staff out in case they have adverse reactions to the shot — such as fever, fatigue or muscle pain — that require them to stay home from work.
That will be even more important when the second dose of the vaccine is administered, three weeks after the first shot; the rate of side effects among clinical trial participants was reported to be higher after the second dose of the vaccine.
Priority will be given to older employees and those who make a request because of medical conditions that leave them especially vulnerable to the coronavirus, Dr. Jarrett said.
No employee will be forced to take the vaccine, Dr. Jarrett said — a decision influenced in part by the fact that the vaccine received an emergency use authorization and has not gone through the F.D.A.’s standard approval process. But he expects demand to be high.
“There will be a certain percentage who are afraid to take the vaccine,” he said. “After they see team members get it and be fine, I suspect most of those will then decide to take it.”
 Ingham County, Mich., which includes Michigan State University, went from reporting about 300 new infections in August to about 1,800 in September.Credit…Sylvia Jarrus for The New York Times
Ingham County, Mich., which includes Michigan State University, went from reporting about 300 new infections in August to about 1,800 in September.Credit…Sylvia Jarrus for The New York TimesAs coronavirus deaths soar across the country, deaths in communities that are home to colleges have risen faster than the rest of the nation, a New York Times analysis of 203 counties where students compose at least 10 percent of the population has found.
In late August and early September, as college students returned to campus and some institutions put into place rigorous testing programs, the number of reported infections surged. Yet because serious illness and death are rare among young coronavirus patients, it was unclear at the time whether the growth of infections on campus would translate into a major health crisis.
But since the end of August, deaths from the virus have doubled in counties with a large college population, compared with a 58 percent increase in the rest of the nation.
Few of the victims were college students but, rather, older people and others living and working in the community.
Since the pandemic began, a Times survey has identified more than 397,000 infections at more than 1,800 colleges and universities. Those cases include more than 90 deaths involving college employees and students.
The link between an outbreak at a college and a coronavirus death in the wider community is often indirect and difficult to document, according to public health experts, especially without extensive contact tracing, which many local health departments in the United States lack resources to pursue.
But researchers have begun finding evidence of ties to college students. Using genetic sequencing to track cases around the University of Wisconsin-La Crosse, Paraic Kenny, a cancer geneticist at the Kabara Cancer Research Institute of the Gundersen Medical Foundation, has found links between infections at the university and cases and deaths in the surrounding region.
That trend highlighted a central fear of health officials — that young adults with limited symptoms might unwittingly transmit the virus, increasing the possibility it would ultimately spread to someone more vulnerable.
Experts suggest an array of ways such spread might happen, including one simple possibility: More than 1.1 million undergraduates work in health-related occupations, census data shows, including more than 700,000 that serve as nurses, medical assistants and health care aides in their communities.
But spread of the virus may also be more invisible, through layers of separation.
“All it really takes is one cavalier interaction,” said Tali Elfassy, an epidemiologist at the University of Miami.
GLOBAL ROUNDUP
 Members of the Public Health Agency of Canada held a drill for the rollout of the coronavirus vaccine in Ottawa on Friday.Credit…Pool photo by Sean Kilpatrick
Members of the Public Health Agency of Canada held a drill for the rollout of the coronavirus vaccine in Ottawa on Friday.Credit…Pool photo by Sean KilpatrickWith the F.D.A.’s authorization of a coronavirus vaccine on Friday, the United States became the third highly developed Western country — after Britain and Canada — to approve such a drug.
But the three countries have different health care systems and face different challenges in the race to get the vaccine to millions of people. Here are some similarities and differences:
For now, yes.
The first vaccine authorized by American regulators, and their British and Canadian counterparts, is the one developed by Pfizer and BioNTech.
But several other vaccines are close behind, particularly one developed by Moderna and the National Institutes of Health, and another from AstraZeneca and the University of Oxford. It could well be that half a dozen or more vaccines are approved in the coming months.
In Britain, very. In the United States, not. In Canada, somewhere in between.
With a strong central government and a National Health Service that covers all of its people, Britain is directing the process from London. The national government chose 50 hospitals that would initially get the vaccine and made sure they were prepared; decided how much each one would receive; and drafted rules determining what order people would be eligible to get it.
In the U.S., the federal government is having the vaccine distributed to each state based on population. It will be up to states to decide how to divide the doses among hospitals, clinics and, ultimately, drugstores and doctors’ offices.
Canada has a universal health care system, but it is decentralized, administered by provinces and territories. For vaccine distribution, the central government plans to work through those regional governments. Ottawa will play a large role in directing the process.
That remains a bit murky.
Canada ordered enough of the Pfizer-BioNTech vaccine for all of its people, Britain enough for 30 percent and the United States enough for 15 percent. But those numbers reflect deliveries that are expected to take months to complete, and Pfizer, like other companies, has hit snags in ramping up production.
All three countries made advance purchases from other companies, as well, so the pace of vaccine approval could significantly affect the speed of rollout. That speed will also be affected by the need for vaccination sites to be equipped with the right freezers, staff, and enough syringes and protective gear.
Initial shipments are a fraction of the pre-purchases — 800,000 doses to Britain and an expected 249,000 this month to Canada. U.S. officials said they hoped to have 40 million doses of the vaccine by the end of the month, which may be optimistic.
In Canada, the government is sending shipments to all 10 provinces. The three northern territories will have to wait. In America, FedEx and UPS will ship vaccines from distribution centers to every part of the country.
That is even murkier.
Britain, Canada and the United States have followed similar strategies, pre-ordering huge numbers of doses — more than enough to inoculate everyone — from multiple makers, hedging their bets in case some of the vaccines are not approved or some manufacturers have production breakdowns.
Relative to their populations, the United States has ordered far fewer doses than Canada or Britain, and last summer it passed up an offer to increase its advance order from Pfizer. Administration officials say the numbers are misleading, because the government has signed options to buy far more of the vaccine if it sees the need.
But in the face of intense global demand, it is not clear how fast pharmaceutical companies will be able to fulfill the orders they have, much less any additional orders.
In other global developments:
In Brazil, the number of people unwilling to take any Covid-19 vaccine has grown to 22 percent, up from 9 percent in August, Reuters reports. President Jair Bolsonaro, a virus skeptic who has continued to critique coronavirus restrictions, said he would not take a coronavirus vaccine. He has been particularly skeptical of China’s Sinovac vaccine. In November, Brazil suspended a late-stage trial of the vaccine. Although Brazil’s cases fell during the fall, there’s been an uptick since early November. It’s now averaging about 43,000 cases a day, about double from a month ago.
Brittanya Karma, a Vietnamese-German rapper and reality television star, died on Nov. 29 in Hamburg, Germany, where she was born and lived. She was 29. The cause was complications of Covid-19, her agent said.
Rizieq Shihab, a radical Indonesian cleric who held large gatherings and called for a “moral revolution” after his return from Saudi Arabia last month, surrendered to the police on Saturday on charges of violating coronavirus protocols. Upon his return, Mr. Rizieq, 55, invited 10,000 people to his daughter’s wedding. Authorities said six of his bodyguards were killed by the police in self-defense. In surrendering, he said he would cooperate with the investigation.
U.S. Roundup
 The Illinois National Guard, at a testing center in May, are assisting with coronavirus testing and screening at the LaSalle Veterans’ Home. At least 38 residents have tested positive for the virus.Credit…Nam Y. Huh/Associated Press
The Illinois National Guard, at a testing center in May, are assisting with coronavirus testing and screening at the LaSalle Veterans’ Home. At least 38 residents have tested positive for the virus.Credit…Nam Y. Huh/Associated PressA coronavirus outbreak at a veterans’ home in Illinois has killed more than a quarter of its residents, prompting the state to mobilize the Illinois National Guard to the home, Gov. J.B. Pritzker said at a news conference on Thursday.
Mr. Pritzker said that 38 residents at the home, LaSalle Veterans’ Home, had contracted the virus out of its 93 total residents, and three staff members were also infected. The governor emphasized that every employee was tested with rapid result antigen tests before each shift, and that residents were being tested every day.
Still, some are criticizing the handling of the outbreak, which was first detected at the home on Nov. 1. Since then, 89 percent of the residents have tested positive for the virus, State Senator Sue Rezin, a Republican, said in a letter on Friday addressed to the veterans at the home. Ms. Rezin said that National Guard medical staff would be assisting with testing and screening, and she noted that the move to mobilize the National Guard was made five weeks after the initial outbreak.
“The fact that this virus was able to run rampant in the facility should be considered unacceptable,” Ms. Rezin wrote in the letter. “With so many unanswered questions remaining, it is critical that the legislature continue its legislative hearings into what happened at the LaSalle Veterans’ Home,” she said.
There are continuing investigations into the outbreak. On Monday, the governor’s office fired the administrator at the LaSalle home and the director of nursing had also been placed on administrative leave. The Illinois Department of Veterans’ Affairs released reports in November, in partnership with the state department of public health and the U.S. Department of Veterans Affairs, detailing several problems in the home.
Alcohol-free sanitizer — which is not effective in eradicating the virus — was stocked in mounted dispensers, a report said. Some staff members were seen in the kitchen area with masks pulled down to their chins and not social distancing. There were staff members wearing gloves, who were seen touching patients and multiple surfaces “without changing or performing hand hygiene,” the report said. “Opportunities for transmission among some staff may have occurred due to reported laxity of masking and social distancing while off duty and also during break periods.”
Illinois has seen a 10 percent decline in cases over the past 14 days, and a 14 percent decline in hospitalizations, after setting seven-day case records at the beginning of November. Yet deaths in the state are up 42 percent in the past two weeks.
The F.D.A. authorized Pfizer’s vaccine for emergency use on Friday, allowing for millions of people to start getting the vaccine within days. Still, the virus continues to decimate the lives of residents at the LaSalle Veterans’ Home, even as residents of nursing homes and long-term-care facilities — and health care workers at high risk of exposure — are at the top of the vaccine waiting list.
In other news around the United States:
Dr. Nicole Alexander-Scott, the director of the Rhode Island’s Department of Health, tested positive for the coronavirus on Saturday. The results came from routine testing, a spokesman for Gov. Gina Raimondo said. Dr. Alexander-Scott was asymptomatic. Ms. Raimondo, who last saw Dr. Alexander-Scott in person during a Covid-19 press briefing on Thursday, is quarantining. She tested negative on Saturday, spokesman Josh Block said. Rhode Island leads the country in coronavirus cases per capita, averaging about 1,240 cases a day this past week, or about 117 cases per 100,000 people. The state has begun issuing temporary licenses to doctors and nurses who are retired, are visiting the state or have recently completed training programs.
As many as 300,000 coronavirus cases across the United States can be traced to a two-day conference in Boston attended by 175 biotech executives in February, according to a study published in the journal Science.
 A vaccination station in Cardiff, England, where the Pfizer and BioNTech vaccine was given to patients this week.Credit…Andrew Testa for The New York Times
A vaccination station in Cardiff, England, where the Pfizer and BioNTech vaccine was given to patients this week.Credit…Andrew Testa for The New York TimesBritish drug regulators recommended on Wednesday that people with severe allergic reactions to food and medicine not receive Pfizer’s coronavirus vaccine after two health care workers, both with such a history, had a serious reaction.
The initial report on the British cases set off alarm and confusion, and left many people with allergies wondering whether the new vaccine would be safe for them.
Here’s what we know so far:
The workers both experienced anaphylaxis, a life-threatening reaction that impairs breathing and can drop blood pressure within minutes or even seconds after exposure to an allergen. Both workers were treated with epinephrine and have recovered, the regulators said.
People with a history of an anaphylactic reaction to any vaccine were excluded from Pfizer’s studies, company officials said on Thursday.
Among those who participated in the Pfizer trials, a very small number of people had allergic reactions. A document published by the F.D.A. on Tuesday said that 0.63 percent of participants who received the vaccine reported potential allergic reactions, compared with 0.51 percent of people who received a placebo.
In Pfizer’s late-stage clinical trial, one of the 18,801 participants who received the vaccine had an anaphylactic reaction, according to safety data published by the F.D.A. None in the placebo group did.
I have allergies, should I be concerned?
Dr. Anthony Fauci, the nation’s leading expert on infectious diseases, said on Wednesday that the allergic reactions were concerning but most likely rare, the kind of effects that show up when a vaccine moves out of testing and into broader distribution.
“If I were a person that had an underlying allergic tendency, I might want to be prepared that I might get a reaction, and therefore be ready to treat it,” Dr. Fauci said, in a webcast moderated by Dr. Sanjay Gupta of CNN, sponsored by Harvard and The New England Journal of Medicine.
Dr. Fauci acknowledged that the problem could turn out to affect a lot of people, but he said that other vaccines would eventually become available for those affected.
Dr. Paul Offit, a vaccine expert at Children’s Hospital of Philadelphia, said the initial, broad recommendation in Britain mentioning severe allergic reactions seemed to be an overreaction that could needlessly scare many people away from a desperately needed vaccine in the middle of a raging pandemic.
Fewer than one in a million recipients of other vaccines a year in the United States have an anaphylactic reaction, Dr. Offit said. Those reactions are treatable and much easier to control than a severe case of Covid-19, he said.
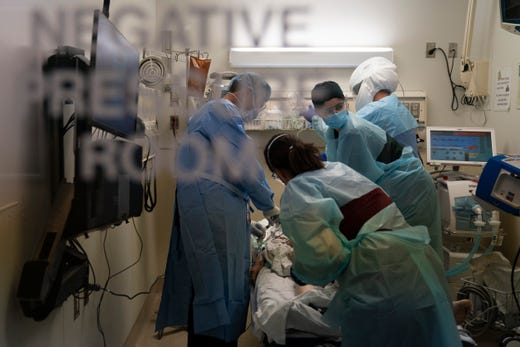
 The stolen ventilators, worth $3 million, were being transported to a Covid-19 intensive care center in El Salvador to treat critically ill patients as part of a federal aid program.Credit…Go Nakamura/Getty Images
The stolen ventilators, worth $3 million, were being transported to a Covid-19 intensive care center in El Salvador to treat critically ill patients as part of a federal aid program.Credit…Go Nakamura/Getty ImagesTwo men have been charged with stealing 192 ventilators owned by the U.S. government in Miami, authorities said on Friday. When they were stolen, the ventilators — worth $3 million — were being transported to a Covid-19 intensive care center in El Salvador to treat critically ill patients as part of a federal aid program.
Yoelvis Denis Hernandez, who is known as Guajiro, and Luis Urra Montero, who goes by the name Flaco, were indicted on charges of federal conspiracy, possessing stolen goods that were being shipped across state lines and the theft of government property.
According to the indictment, Mr. Hernandez, 42, and Mr. Montero, 24, stole a tractor-trailer in August that was being used to transport the ventilators to Miami International Airport. The tractor-trailer had been left in a lot overnight. After an investigation, most of the ventilators were found, the authorities said.
At the onset of the pandemic, medical professionals and government authorities were concerned about the shortage of ventilators in the United States and in other countries. Since then, however, the country’s medical device manufacturers have ramped up production of the machines. The U.S. now has more than 200,000 critical care ventilators — 155,000 of them in the Strategic National Stockpile — but there aren’t enough specialists to operate them.
In June, a man was charged with stealing ventilators from a Veterans Affairs medical center in Seattle and selling them on eBay.
as per our monitoring this Story originally appeared * : ) here → *
Covid-19 Updates: First U.S. Vaccines Are About to Be Shipped as Virus Ravages America
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

Teva to be Sole Distributor of COVID-19 Vaccines in Israel
Photo Credit: Yonatan Sindel / Flash 90

 TEVA Pharmaceutical Industries headquarters in Jerusalem
TEVA Pharmaceutical Industries headquarters in Jerusalem The Israeli Teva Pharmaceuticals firm has signed an exclusive agreement with the government to distribute all the COVID-19 vaccinations in the Jewish State.
Teva Israel CEO Yossi Ofek told the Globes business news site on Sunday, “We are waiting for the green light and the possibility that within several weeks millions of vaccination shots will arrive here.
Advertisement <!–Publisher #16: JewishPress.com Zone #113: Comment Banner / (02) / News Size #15: Banner 468×60 (Comments and Mobile) [468×60] –>
“It’s looking like Israel will be the first place after the US that will receive vaccinations for a large part of the population, even before Britain.
“But since the announcement the vaccine shots are set to arrive in larger numbers than expected. We have strengthened the readiness at TEVA SLA’s logistics center in Shoham and Teva’s center in Kfar Saba.
“On both these sites there are already freezers that can chill Pfizer’s vaccinations to minus 70 degrees centigrade, and we are in the process of obtaining more freezers, so that within several weeks we can take in three million vaccination doses at this temperature. Moderna’s vaccination is kept at a less low temperature and we are already prepared to receive them today,” he said.
“This is something that we have not ever previously experienced in this field,” Ofek underlined. “We have developed methods in the country to cope with it, from the moment that the plane leaves the factory to us. The process is very complicated and has to be 100 percent successful.”
The plan entails distributing the vaccine to hundreds of clinics, care homes and possibly other institutions as well. At these destinations, the HMO takes responsibility for the package, but from the moment the mini-package comes out of the freezer, it can be kept at the typical temperature for vaccinations of 2 to 8 degrees centigrade for up to five days only. In each package there will be 970 doses, meaning every package will hold enough for 970 Israelis to be vaccinated.
The Health Ministry is expected to give the green light to vaccinate Israel’s population as soon as the FDA approves the vaccine, Ofek said.
“We have responsibility for any defect in the product during the period it is in our hands,” he told Globes. “Regarding responsibility for the product itself, for the efficacy and safety of it, these are part of agreements between Israel and Pfizer and Moderna, in which we are not involved.”
Advertisementas per our monitoring this Story originally appeared * : ) here → *
Teva to be Sole Distributor of COVID-19 Vaccines in Israel
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

Coronavirus updates: California region has 0.0% available ICU capacity; US reports 16 millionth case; vaccinations to begin Monday
CLOSE![]()
The COVID-19 vaccine is using new technology that has never been used before in traditional vaccines. Here’s how an mRNA vaccine works. USA TODAY
The United States on Saturday reported its 16 millionth COVID-19 case, even as a newly authorized vaccine is already being distributed and vaccinations are expected to begin Monday, according to federal officials.
It took three months for the U.S. to record its first million cases. But it’s been just four days between the 15th and 16th million case milestones — yet another sign the virus is spreading at an alarming and deadly pace. But the nation’s first authorized vaccine promises to help slow the pandemic in coming months, as the federal government races to quickly distribute it.
“As I speak today, right now, vaccines are being packaged,” Gen. Gustave Perna, co-leader of Operation Warp Speed in charge of logistics, said Saturday. “Tomorrow morning, vaccines will start rolling from manufacturing to distribution hubs. By Monday, vaccines will be received.”
Vaccinations will begin Monday, Dr. Nancy Messonnier, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during a meeting Saturday.
Health care workers and nursing home residents are first in line. Patients and caregivers will receive fact sheets describing the risks and benefits, she said.
Earlier Saturday, Food and Drug Administration officials reassured the American public that the agency did not cut corners in its review and urged people to get vaccinated. The United States is on the cusp of losing 300,000 people to COVID-19.
Keep refreshing this page for the latest updates. For headlines in your inbox, sign up for our newsletter, Coronavirus Watch.
Here are today’s top headlines:
- An advisory committee to the Centers for Disease Control and Prevention voted Saturday to recommend the Pfizer/BioNTech vaccine for people 16 and older. Now it goes to the director of the CDC for his signature.
- The Food and Drug Administration late Friday granted emergency authorization to Pfizer/BioNTech’s vaccine that research shows is highly effective at preventing a disease.
- The promising news will not immediately end the pandemic, which is raging out of control. Dr. Ugur Sahin, CEO of BioNTech, estimated it would be March or April before the vaccine could impact the virus’ spread — underscoring the need for measures like mask-wearing and social distancing in the meantime.
- About 1 in 8 U.S. hospitals had few or no intensive care unit beds available last week, according to new federal data. Experts say the number of hospitals struggling to accommodate the nation’s sickest patients likely will increase following another week of record COVID-19 cases.
- Two Florida men are facing federal charges that allege they stole nearly 200 ventilators the United States shipped to El Salvador to help the Central American country treat coronavirus patients.
Another day of record deaths in the US: On Friday, 3,309 people in the U.S. died from the coronavirus, according to Johns Hopkins University data. The previous record was set Wednesday at 3,124, the first time the daily death toll surpassed 3,000. As of Saturday, about 297,000 people in the U.S. have died, with more than 16 million confirmed cases. The global totals: 71.6 million cases and 1.6 million deaths.
What we’re reading: We’re answering your questions about the vaccine, like: What are the side effects? Can you still get sick? Is it safe during pregnancy? Read more here.
New York’s 21 Club closes indefinitely due to pandemic
The storied 21 Club in midtown Manhattan, a favorite of celebrities and the power elite for nine decades, is closing indefinitely due to the coronavirus pandemic, but the owners are optimistic about reopening at some point.
The restaurant’s owners filed notice about the closing with the city on Wednesday, saying all 148 employees will be terminated on March 9. The news comes as indoor dining at New York City restaurants will be banned again starting Monday in an effort to halt a resurgence of the virus, but takeout and outdoor dining will still be allowed.
The restaurant has been closed since last March during the first wave of the pandemic, as indoor dining was prohibited.
The 21 Club was a favorite dining place of presidents since Franklin D. Roosevelt’s day. Shortly after the 2016 election, then-President-elect Donald Trump broke protocol by leaving his Trump Tower residence without taking along his press contingent to eat at the restaurant. John Steinbeck, Ernest Hemingway and Frank Sinatra had favorite tables at 21, and Humphrey Bogart proposed to Lauren Bacall there. It appeared in films ranging from the 1950 Bette Davis classic “All About Eve” to 1987’s “Wall Street.”
— Associated Press
12-county California region has 0.0% available ICU capacity
In San Joaquin County, part of California’s vast Central Valley that produces most of the country’s fruits and vegetables, the coronavirus is spreading like a weed and the hospitals are running out of beds for the sickest patients.
San Joaquin is part of a 12-county region that on Saturday, according to the California Department of Public Health, had 100% of its intensive care unit beds filled, the highest rate anywhere in California. And with cases continuing at an unprecedented rate, the death toll inevitably will grow, too.
A new stay-at-home order was imposed this week but it is unknown whether it will have the intended consequence of finally changing enough people’s behavior to slow infections as a vaccine is widely rolled out.
— The Associated Press
UPS Worldport hub in Louisville to soon ship vaccine
The first shipments of Pfizer/BioNTech’s COVID-19 vaccine will move through UPS’ Worldport global air hub at the Louisville, Kentucky, airport Sunday, a company spokesman told The Courier Journal, part of the USA TODAY Network.
UPS said the vaccine will originate from storage sites in Michigan and Wisconsin.
“The vaccines will be transported to UPS Worldport facilities in Louisville, where they will be expedited Next Day Air to select destinations, including hospitals, clinics and other medical facilities, to inoculate healthcare workers,” the company said in a news release.
The shipments will arrive at the hub to be sorted and will be shipped out — all on Sunday, UPS spokesman Jim Mayer said. He said he did not know how many doses will be moving through. Mayer previously said that UPS will deliver to states in the eastern half of the United States. FedEx will deliver to the western half of the U.S.
– Ben Tobin, Louisville Courier Journal
‘We are not taking a victory lap,’ Operation Warp Speed official says
The chief operating officer of Operation Warp Speed, the White House effort to fund, produce and distribute a vaccine, cheered the emergency authorization of the Pfizer/BioNTech vaccine but warned of a tough road ahead.
“While this moment is extraordinary … we have a lot of work to do. We are not taking a victory lap,” Perna said Saturday.
He said it wouldn’t have made a difference to Operation Warp Speed if the FDA had authorized the vaccine Saturday instead of Friday. “Our ultimate goal was to get it there no later than Monday morning,” he said.
Perna reiterated that Operation Warp Speed wouldn’t distribute the second vaccine dose or a portion held in reserve until it has “ultimate confidence” that there are enough doses and the delicate distribution system works. That could change in January or February.
“We want no vaccines on a shelf,” Perna said. “Doesn’t matter how effective a vaccine is if it’s on a shelf.”
Perna said vaccine doses were not positioned in advance because the operation “did not want to presume” emergency use authorization. “Under no circumstances did we want to get ahead of the great FDA and their decision making,” he said.
FDA: No corners cut in reviewing Pfizer/BioNTech vaccine
Amid concerns about how quickly the FDA issued an emergency authorization for the Pfizer/BioNTech vaccine, agency leaders stressed Saturday that they conducted a thorough, transparent review.
“Science and data guided the FDA’s decision. We worked quickly because of the urgency of this pandemic, not because of any other external pressure,” FDA Commissioner Stephen Hahn said. “I will absolutely take this COVID-19 vaccine.”
Hahn said reports Friday that the White House had threatened to fire him if the agency did not authorize the vaccine were “inaccurate.”
Hahn said the agency was “very concerned about vaccine hesitancy” and made the process as transparent as possible by posting trial data and documents online.
“Efficiency does not mean any cutting of corners,” Hahn said. The FDA “found ways to cut the red tape,” but “important safety checks remained in place,” he said.
Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said the FDA requires vaccine developers to monitor for “any significant adverse events” as they seek standard approval. The FDA and the CDC are monitoring as well, he said.
Officials provided more information about whether certain groups of people should get the vaccine. People who are pregnant or immunocompromised, who were not included in safety trials, should discuss the vaccine with their providers “on an individual basis,” Marks said.
Vaccination sites will be equipped to treat allergic reactions, official says
Sites where the Pfizer/BioNTech vaccine are administered will be able to treat allergic reactions, Marks said. About 1.6% of the population has had a severe allergic reaction to food or something in the environment but are eligible to receive the vaccine, Marks said. Only people who have had a severe allergic reaction to a previous dose of the vaccine or one of its components should not receive the vaccine.
Experts say the ingredients in the Pfizer/BioNTech vaccine look typical: an active ingredient (in this case, messenger RNA), fats, salt and sugars.
Dr. Matthew Heinz, a hospitalist based in Tucson, Arizona, said if any part of the vaccine were to trigger an allergic reaction, it could be one of the components of the fat molecule. But that’s rare. “We’re talking about a number of relatively mild reactions that you can count on one hand,” out of tens of thousands of people in the study, he said.
Two British people with severe allergies apparently had reactions to Pfizer/BioNTech’s vaccine this week. Allergic reactions were not a significant problem in the U.S. trial, in which more than 20,000 people received two doses of the vaccine. The U.S. trials kept out subjects who have had severe allergic reactions.
— Adrianna Rodriguez
Friday set another record for COVID-19 cases, deaths
On the same day that the FDA approved the first vaccine for emergency use in the United States, the nation reached another milestone in COVID-19 cases and deaths.
The country recorded 3,309 coronavirus deaths Friday, according to Johns Hopkins University data, surpassing Wednesday’s record when 3,124 people died. Friday also saw the highest number of daily cases to date in the U.S.: 231,775.
Pfizer’s COVID-19 vaccine gets FDA authorization
In what is hoped to be the beginning of the end of America’s COVID-19 pandemic, the FDA on Friday evening authorized the first vaccine to prevent people from getting sick.
What this means: The Pfizer/BioNTech vaccine received emergency use authorization, which is not full approval. Although it has received all the standard short-term safety and effectiveness reviews, the vaccine has not been tested for the two years typical of an approved vaccine. So it is not yet clear how long protection will last.
When will you get it? Frontline health care workers and nursing home residents are expected to get the vaccine first. More doses will be rolled out in the weeks and months to come, with Pfizer and Moderna each expected to deliver 100 million doses of their vaccines by the middle of next year.
What about other vaccines? Next week, a similar COVID-19 vaccine developed by the Massachusetts-based Moderna will go through the same review process, and could swiftly be cleared for use.
– Karen Weintraub
In America’s hardest-hit county, COVID deaths claimed parents, friends
Just over a month ago, the coronavirus pandemic tore through Quinter, Kansas, a rural town of 1,000, killing 20 residents. Here, where most everyone knows most everyone else, the pandemic has killed farmers and their wives. The town’s unofficial historian. The beloved grandmother whose sour cream chocolate cake with chocolate fudge frosting was always the talk of the party. The mom whose piano-playing still echoes in the heads of her friends.
And it has drained the hearts of the survivors. Those who feel guilty about recovering. The ambulance workers battling to treat their own relatives. The exhausted doctor who watched nearly half his patients die.
As of Thursday, the coronavirus has killed a higher percentage of Gove County residents than any other county in the United States: one out of every 132 people. Even today, mask-wearing remains controversial. Friendships are strained as authorities struggle to persuade their neighbors to follow basic public health guidelines, such as avoiding large gatherings.
“We are living through history right now, and I worry what the history books will say about us,” said Ericka Nicholson, 47, who helps run the town’s volunteer ambulance service and survived the infection. Read more from Gove County, Kansas.
– Trevor Hughes
CLOSEWe asked you to tell us your biggest questions about the COVID-19 vaccines. Here are some answers. USA TODAY
Relentless coronavirus surge fills 1 in 8 hospital ICU units
About 1 in 8 U.S. hospitals had little or no intensive care unit space available last week. Experts say the number of hospitals struggling to accommodate the nation’s sickest patients likely will increase after another week of record COVID-19 cases.
The federal government this week released a sweeping database showing a one-week average of COVID-19 patients in hospitals nationwide. It’s the first time the Department of Health and Human Services has provided such detailed information on nearly 5,000 U.S. hospitals since the pandemic began.
The absence of hospital-level information has been a blind spot in the pandemic, as administrators must call neighboring hospitals to transfer patients when they run out of beds and staff.
“We know what’s happening in our system, but I don’t know what’s happening in the other systems,” said Dr. Lewis Kaplan, professor of surgery at University of Pennsylvania Hospital in Philadelphia. “To have a coordinated effort you need to have that kind of data so everyone knows where everyone else happens to be.” Read more.
– Ken Alltucker and Aleszu Bajak
US buys another 100M doses of Moderna’s vaccine
The Trump administration announced Friday that it has purchased an additional 100 million doses of a coronavirus vaccine made by Moderna, bringing the federal government’s total order from the company to 200 million doses. The additional doses would “provide for continuous delivery through the end of June 2021,” the Department of Health and Human Services said in a press release.
“Securing another 100 million doses from Moderna by June 2021 further expands our supply of doses across the Operation Warp Speed portfolio of vaccines,” HHS Secretary Alex Azar said in a statement. “This new federal purchase can give Americans even greater confidence we will have enough supply to vaccinate all Americans who want it by the second quarter of 2021.”
Moderna became the second vaccine maker in the U.S. to request authorization from the FDA at the end of last month. The company’s latest findings showed that of 196 people in the clinical trial who caught COVID-19, 185 of them had received the placebo, while only 11 had received the vaccine. That works out to an effectiveness rate above 94%.
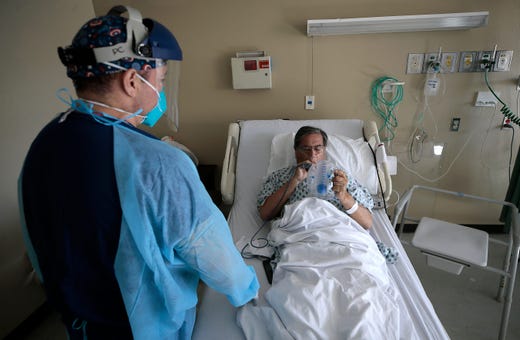
Wisconsin woman reunites with family after battling virus for nearly 3 months
After spending over 80 days battling COVID-19 in a Wisconsin hospital, Nancy Van Dyn Hoven on Friday was reunited with her family and friends — just in time for the holidays.
“Other than 2020 being awful, this is just the best,” Van Dyn Hoven, 60, said Friday at home, chuckling with her husband, Dennis, and daughter, Stacy Arnoldussen. “It’s all I could ask for.”
Dr. Anthony Zeimet, an infectious diseases specialist at the hospital and Nancy’s doctor, said the severity of her illness was particularly surprising because she was just 59 years old — she turned 60 in the hospital — and had no underlying health conditions that would put her at higher risk of severe illness.
“The virus kind of ravaged her body,” Zeimet said. “It just goes to show that, with COVID, we don’t know who’s going to do well or do poorly. Nancy was someone who, when she was first admitted, we thought she’d do pretty well. … Unfortunately, she ended up being here for 80-plus days.”
Although Nancy’s recovery is far from over and she has a long road of rehabilitation services ahead, Friday marked the end of a months-long struggle with the virus. .
— Samantha West, Appleton Post-Crescent



















































Artists Jack Schwab, and Debbie Wilger, wear their masks July 14, 2020, inside the Missouri Artists on Main store in downtown St. Charles, Mo. Schwab, 60, who makes silver jewelry, and Wilger, 63, a painter, are concerned about the uptick in coronavirus cases in St. Charles County, and say most customers in the store abide by their facial covering policy, but a few have left in anger because of it. Jim Salter, AP






























Alice Mayes, 92, is visited by her family at Signature HealthCARE on May 6, 2020 in NewBurgh, Ind. The family, from left, Onya Rhoades, Lexi Rhoads, 3, Dylan Rhoades, 5, Kaitlyn Helmbrecht, 2, James Helmbrecht and Del Mayes were separated by a window glass on May 6, 2020 in Newburgh, Ind. The 92-year-old is a COVID-19 survivor. Denny Simmons, Evansville Courier & Press































Austin High School seniors and best friends, clockwise from top left, Brooke Peterman, 17, Maddy McCutchin, 18, Lucia Saenz, 17, Reese Simek, 18, and Lily Tickle, 18, visit with each other in the parking lot at the school in Austin, Texas, on Sunday April 5, 2020. In the midst of a shelter in place order due to the coronavirus pandemic, the girls sat in the back of their cars to chat at a safe distance. Jay Janner, Austin American-Statesman / USA TODAY Network








Replay
Autoplay
Show Thumbnails
Show Captions
Read or Share this story: https://www.usatoday.com/story/news/health/2020/12/12/covid-vaccine-update-pfizer-vaccine-authorized-first-doses-coming/3893846001/
as per our monitoring this Story originally appeared * : ) here → *
Coronavirus updates: California region has 0.0% available ICU capacity; US reports 16 millionth case; vaccinations to begin Monday
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic
Add Story/ Your Say Until the Cause is Concluded
[*Select Category/Tag:
Coronavirus Covid-19 Pandemic
at Your Next Publish Screen.]

Anti-vaccine figure partners with Roku after YouTube banned him for sharing dangerous coronavirus misinformation
Anti-vaccine figure partners with Roku after YouTube banned him for sharing dangerous coronavirus misinformation

Broadcast and streaming platform Roku has added an online show from anti-vaccine activist Del Bigtree, who has repeatedly encouraged people to intentionally contract COVID-19. The program is listed as “educational” by Roku. In late July, YouTube removed Bigtree’s The HighWire channel for violating the platform’s policies after Media Matters reported on Bigtree using YouTube to spread dangerous medical misinformation.
During The HighWire’s latest broadcast, which aired on August 27 on Roku and also on his other biggest streaming platform, Facebook, he again argued that people should seek to contract COVID-19 in order to build natural herd immunity, a course of action that would lead to millions of deaths in the U.S.
Bigtree, who has no medical credentials, is a leading figure in the anti-vaccination movement through his anti-vaccine nonprofit organization Informed Consent Action Network and as the host of The HighWire, which is broadcast on Thursdays. A 2019 profile of Bigtree in the online parenting magazine Fatherly labeled him “dangerous” and said he “may be the most connected node in the anti-vaccine activist network.”
On August 19, The HighWire’s Twitter account teased the show’s “brand new ROKU Channel.” In an August 27 email to supporters, Bigtree wrote that following the YouTube ban, “we have started establishing relationships with companies who share in our vision of providing a new space for you to get real news, real science, transparent, and uncensored” and as part of that effort “we’ve also launched The HighWire on Roku!” Roku prohibits channel owners from publishing content that “place[s] individuals or groups in imminent harm” or material found to “contain false, irrelevant or misleading information.”
Bigtree’s Roku channel offers episodes dating back to July 30. During the July 30 broadcast, which was largely about his channel being banned from YouTube, Bigtree defended his claim that people should “catch this cold” by intentionally contracting COVID-19 “to create herd immunity” and said it was “incredible” that he was able to continue to broadcast live on Facebook.
In contrast to YouTube’s outright ban of Bigtree’s channel, Facebook — where Bigtree has over 348,000 followers — has taken a more piecemeal approach to containing his promotion of conspiracy theories and dangerous medical misinformation. Following Media Matters’ reporting, Facebook removed some videos where Bigtree made dangerous claims, but not others that contained similar dangerous claims. Facebook applied a fact-checking label to his August 27 broadcast — to note that Bigtree’s suggestion that the novel coronavirus was manmade is a conspiracy theory — but took no action regarding the dangerous claims he made during the broadcast that people should intentionally contract COVID-19 to build natural herd immunity.
During that broadcast, which is available on Bigtree’s new Roku channel, Bigtree argued that Americans should be willing to assume the same level of risk of dying of COVID-19 as we would if “China was attacking our borders.” Along those lines, Bigtree said, “I assure you, we would risk the lives of millions.” Bigtree then argued that the maximum number of Americans who could die from COVID-19 was “just nearing 200,000 total casualties, people who most likely were probably going to die this year anyway.” In fact, more than 183,000 Americans already have died of COVID-19 (and the actual death toll, based on total excess deaths, is even higher) and there is no end in sight to hundreds of Americans dying of the disease each day.
as per our monitoring this Story originally appeared * : ) here → *
Anti-vaccine figure partners with Roku after YouTube banned him for sharing dangerous coronavirus misinformation
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

‘Mix-and-match’ coronavirus vaccines to be tested
‘Mix-and-match’ coronavirus vaccines to be tested
Ms Keenan, who is originally from Enniskillen, said: “I feel so privileged to be the first person vaccinated against Covid-19. It’s the best early birthday present I could wish for because it means I can finally look forward to spending time with my family and friends in the new year after being on my own for most of the year.”
as per our monitoring this Story originally appeared * : ) here → *
‘Mix-and-match’ coronavirus vaccines to be tested
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

Covid-19 Updates: First U.S. Vaccines Are About to Be Shipped as Virus Ravages America
Covid-19 Updates: First U.S. Vaccines Are About to Be Shipped as Virus Ravages America
Last Updated
Dec. 13, 2020, 11:50 p.m. ET
Dec. 13, 2020, 11:50 p.m. ETWith Pfizer’s vaccine cleared for emergency use on Friday, states eagerly began their own preparations to receive the vaccine within days. Deaths are rising sharply in college towns.
This briefing has ended. Follow our latest coverage of the coronavirus pandemic.
Here’s what you need to know:
UPS and FedEx say plans to ship the vaccine are underway.
A C.D.C. panel has endorsed the Pfizer vaccine for people 16 and over.
With the first injections expected to be given as early as Monday, this is how states are preparing.
Charley Pride, country music star, dies from complications of Covid-19.
The largest health care provider in the New York region is frantically prepping for the arrival of the vaccine.
Deaths from the virus have risen sharply in college towns — but few have been college students.
Britain, Canada and the U.S. start their vaccination campaigns, and other news from around the world.
An outbreak at an Illinois veterans home has killed more than a quarter of residents.
 UPS said it would transport doses of the vaccine from storage sites in Michigan and Wisconsin to its air cargo hub in Louisville, Ky.Credit…John Sommers Ii/Reuters
UPS said it would transport doses of the vaccine from storage sites in Michigan and Wisconsin to its air cargo hub in Louisville, Ky.Credit…John Sommers Ii/ReutersUPS and FedEx, normally rivals, are working side by side to ship the coronavirus vaccine developed by Pfizer and BioNTech, the first of the vaccines to win U.S. government approval.
The two shipping companies said they had put plans they had been working on for months into action after the Food and Drug Administration gave the vaccine emergency authorization late Friday.
Delivery of the first vaccines comes as the virus continues to rage across America, with officials reporting more than 207,000 new cases on Saturday. That brought the total number to more than 16 million, by far the most in the world, less than a week after the country surpassed 15 million. More than 3,000 deaths were reported for the first time on Wednesday, and the country’s total is approaching 300,000.
At a news conference on Saturday, Gen. Gustave F. Perna, the chief operating officer of Operation Warp Speed, the federal effort to bring a vaccine to market, said that boxes were being packed at Pfizer’s plant in Kalamazoo, Mich., and would be shipped to UPS and FedEx distribution hubs, where they would be dispersed to 636 locations across the country. Pfizer said shipping would start early Sunday morning.
Mr. Perna specified that 145 sites would receive the vaccine on Monday, 425 on Tuesday and 66 on Wednesday.
“Make no mistake, distribution has begun,” he said. UPS said it expected to start transporting the vaccine on Sunday morning, when employees stationed at Pfizer’s facility in Michigan will affix special Bluetooth- and radio-enabled tracking tags to each shipment. An aircraft waiting nearby will take the vaccine to the company’s Worldport air cargo hub in Louisville, Ky., a sprawling 5.2-million-square-foot sorting facility. Future shipments of the vaccine will also be transported by truck to the Louisville hub, which is up to a six-hour drive from Michigan.
“This is the moment we’ve been waiting for,” Wes Wheeler, president of the company’s health care division, said in an interview on Saturday. “We’ve been planning for months with daily calls, drilling down to really quite minute details.”
Mr. Wheeler said he planned to oversee the first vaccine shipments from a newly established 24/7 command center in Louisville, which will be staffed at any given time by teams of five to 10 people who will monitor each vaccine package as it moves through the UPS network.
Once the vaccine shipments arrive at the Louisville hub, they will be sorted alongside other packages and distributed to hospitals and other medical facilities. Every truck driver or airplane pilot will know if they are carrying a vaccine package, Mr. Wheeler said. Both UPS and FedEx have said that doses will arrive at their destinations a day after leaving the Pfizer facilities.
Even before the vaccine was approved, UPS had already started shipping out kits with the medical supplies needed to administer it, such as alcohol wipes and syringes, Mr. Wheeler told a Senate subcommittee this week. UPS and FedEx will split distribution of the vaccine throughout the country. After those shipments arrive, all Pfizer dosing sites will receive another shipment from UPS of 40 pounds of extra dry ice to keep the vaccines at a frigid temperature, he said.
“You have two fierce rivals here, and competitors, in FedEx and UPS, who literally are teaming up to get this delivered,” Richard Smith, a FedEx executive, told the Senate’s Subcommittee on Transportation and Safety on Thursday.
Both companies said the shipments would be closely tracked and monitored, and would be given priority over other packages. To ship its vaccine, Pfizer designed specialized containers packed with enough dry ice to keep a minimum of 975 doses cool for up to 10 days. Each comes with a tracking device.
Like UPS, FedEx said it would also affix its own tracking tags to vaccine shipments. Each UPS truck carrying the doses will have a device that tracks its location, temperature, light exposure and motion, Mr. Wheeler told the senators. The company’s trucks will have escorts, too, he said on Thursday. It is not clear whether he meant the local police or other government officials, or possibly private guards, and he declined to elaborate on that and other details in the interview, citing security concerns. But the trucks leaving Pfizer’s facility will be tracked “by the minute,” he said.
 Students at the Mast Academy in Miami arriving for classes in October. The vaccine has been approved for use in 16- and 17-year-olds.Credit…Lynne Sladky/Associated Press
Students at the Mast Academy in Miami arriving for classes in October. The vaccine has been approved for use in 16- and 17-year-olds.Credit…Lynne Sladky/Associated PressAn independent committee of experts advising the Centers for Disease Control and Prevention on Saturday afternoon recommended the Pfizer coronavirus vaccine for people 16 and older. That endorsement, which now only awaits final approval by Dr. Robert Redfield, the director of the C.D.C., is a key signal to hospitals and individual health care providers that they should proceed to inoculate patients.
It follows Friday night’s emergency use authorization of the vaccine by the Food and Drug Administration, which oversees licensing of medical products.
“Based on everything we saw from looking at the data, we’re very comfortable that the safety profile that was observed in 17- and 16-year-olds was acceptable,” Dr. Peter Marks, the F.D.A.’s top vaccine regulator, said at a news conference on Saturday.
The C.D.C. advisory committee, which typically meets three times a year to review amendments to routine schedules for child, adolescent and adult vaccines, has been engaged in numerous marathon-length sessions this fall to discuss a plethora of issues surrounding the introduction of limited supplies during a pandemic of the novel vaccine.
In meetings on Friday and Saturday, the panel’s heated discussion centered mainly on three areas: whether to recommend the vaccine for patients 16 and 17 years old, for pregnant and lactating women, and for patients who have had an anaphylactic reaction to other vaccines.
C.D.C. officials and scientists will review the debate and post more precise guidance about those specific groups and others on Sunday and throughout next week, as more information about the vaccine becomes known.
 Vail Health Hospital placed mock coronavirus vaccines into the hospital’s ultracold freezer on Tuesday in Colorado.Credit…Helen H. Richardson/The Denver Post, via Associated Press
Vail Health Hospital placed mock coronavirus vaccines into the hospital’s ultracold freezer on Tuesday in Colorado.Credit…Helen H. Richardson/The Denver Post, via Associated PressThis weekend, 2.9 million doses of the Pfizer and BioNTech vaccine are to begin traveling by plane and guarded truck from facilities in Michigan and Wisconsin to designated locations, mostly hospitals, in all 50 states.
The first injections are expected to be given by Monday to high-risk health care workers, the initial step toward the goal of inoculating enough Americans by spring to finally halt the spread of a virus that has killed nearly 300,000, sickened millions and upended the country’s economy, education system and daily life. There are now more than 16 million virus cases reported in the United States, according to a New York Times database.
The rapid development of the vaccine, and the F.D.A.’s emergency authorization of it on Friday night based on data showing it to be 95 percent effective, has been a triumph of medical science, but much in this complicated next stage could go wrong.
States say they have only a fraction of the funding they need from the federal government for staffing to administer the shot, for tracking who has received both doses of the vaccine — a booster is needed three weeks after the initial injection — and for other crucial pieces of the effort.
But for all the planning that has been done and contingencies that have been put in place in recent months, there is still a good deal of confusion. States are receiving initial allocations according to a federal formula based strictly on their adult population, but many hospitals say they still don’t know exactly how much they will get or when shipments will arrive. Some hospital systems are reeling from the news that their initial allocations will be much smaller than they had hoped.
One reason for the shortfall in initial supply is that federal officials decided to send out fewer than half of the 6.4 million doses they had planned for the first wave.
Although there is some variation among their plans, states are largely planning to follow recommendations from the Centers for Disease Control and Prevention about who gets vaccinated first: health care workers at high risk of exposure to the virus and residents of nursing homes and other long-term-care facilities, a population that has died from the virus at disproportionately high rates.
On Thursday, as an F.D.A. advisory committee debated whether to recommend authorization of the Pfizer vaccine, the first packages of supplies to administer it — vaccination record cards, masks, visors, information sheets and syringes — arrived at UPMC Presbyterian, a hospital in Pittsburgh.
 Charley Pride performing in New York in 1975.Credit…Bettmann Archive, via Getty Images
Charley Pride performing in New York in 1975.Credit…Bettmann Archive, via Getty ImagesCharley Pride, who was celebrated as a country music’s first Black superstar and known for hits such as “Kiss an Angel Good Mornin’” and “Is Anybody Goin’ to San Antone,” died on Saturday at age 86 while in hospice care in Dallas.
The cause of his death was complications of Covid-19, said Jeremy Westby, the singer’s publicist.
Lauded as a star who paved the way for other country music artists of color, Mr. Pride was praised for his contributions to the country music canon. In November, he received the Country Music Association’s Lifetime Achievement Award at the 54th annual Country Music Association Awards, where he also performed.
Some guests at the show, which was Mr. Pride’s last public event, were not wearing masks. However, event organizers said all protocols from the Centers for Disease Control and Prevention were followed.
Born March 18, 1934 in Sledge, Miss., Mr. Pride served in the army before moving to Montana to try to make it as a baseball player. There, he worked at a smelting plant and played semiprofessional baseball in East Helena, where he was paid $10 to sing the national anthem before games.
He began his country music career in the 1960s after signing with RCA Records, later quitting his factory job after his 1967 song “Just Between You and Me” became a Top 10 country hit.
As a Black man entering the country music industry in the 1960s, Mr. Pride’s career did not escape prejudice. Once radio stations learned he was Black, many refused to play his music, and during a 1970s award show, singer Loretta Lynn was told not to embrace him should he win an award she was presenting. (She did so anyway.)
“We’re not colorblind yet,” Mr. Pride wrote in his memoir, “Pride: The Charley Pride Story” (1994). “But we’ve advanced a few paces along the path, and I like to think I’ve contributed something to that process.”
During his career, Mr. Pride recorded over 50 hits that made it to Top 10 on the country charts, with over 20 hitting No. 1. Mr. Pride also racked up notable awards: Country Music Association’s male vocalist of the year in 1971 and 1972, Country Music Association’s entertainer of the year in 1971 and a lifetime achievement Grammy Award in 2017.
Dolly Parton said on Saturday she was “heartbroken” over Mr. Pride’s death, calling him one of her “dearest friends.”
“What a horrible, horrible virus,” she said on Twitter. “Charley, we will always love you.”
Mr. Pride is survived by his wife, Ebby Rozene Cohran Pride, and his children, Carlton, Charles and Angela.
 Freezers at Northwell Health at New Hyde Park, N.Y., awaiting doses of the coronavirus vaccine.Credit…Northwell Health
Freezers at Northwell Health at New Hyde Park, N.Y., awaiting doses of the coronavirus vaccine.Credit…Northwell HealthThe emergency authorization of the Pfizer-BioNTech vaccine on Friday night has set off a frenzy of preparations at hospitals and doctors’ offices across the United States. Among them is Northwell Health, the largest health care provider in the New York region, once the coronavirus epicenter in the United States.
Organizing the receipt and further distribution of the vaccine is a mammoth task. Northwell has set up nine vaccination centers equipped with ultracold freezers at or near its hospitals in Manhattan, Staten Island, Westchester County and Long Island, and plans to start vaccinating tens of thousands of employees as soon as the vaccine shipments arrive, probably on Monday, said Dr. Mark Jarrett, Northwell’s chief quality officer.
The health system, which has 56,000 employees who interact with patients, has already received shipments of kits with supplies for administering the vaccine, including syringes, alcohol swabs and gloves.
Northwell expects to have all of its workers — not just care providers like doctors and nurses, but also others such as cleaning personnel who work in intensive care units and patient rooms — vaccinated within five weeks with the Pfizer-BioNTech or the Moderna vaccines, Dr. Jarrett said.
Employees will be given appointments to come in for shots so they won’t have to stand in line and can maintain social distancing. A backup list of additional recipients will be drawn up in case anyone has to cancel an appointment at the last minute.
“The one thing we don’t want to do is ever waste vaccine,” Dr. Jarrett said. The Pfizer-BioNTech vaccine, which is transported on dry ice, has to be stored at minus 94 degrees Fahrenheit, and then prepared for use.
“Once you’ve defrosted and diluted it, you only have six hours to use it,” Dr. Jarrett said. “You want to certainly use every dose that’s in a vial.”
The health system is carefully scheduling employees for vaccination in various units and hospitals to prevent staffing problems from overburdening any single unit. The idea is to evenly spread staff out in case they have adverse reactions to the shot — such as fever, fatigue or muscle pain — that require them to stay home from work.
That will be even more important when the second dose of the vaccine is administered, three weeks after the first shot; the rate of side effects among clinical trial participants was reported to be higher after the second dose of the vaccine.
Priority will be given to older employees and those who make a request because of medical conditions that leave them especially vulnerable to the coronavirus, Dr. Jarrett said.
No employee will be forced to take the vaccine, Dr. Jarrett said — a decision influenced in part by the fact that the vaccine received an emergency use authorization and has not gone through the F.D.A.’s standard approval process. But he expects demand to be high.
“There will be a certain percentage who are afraid to take the vaccine,” he said. “After they see team members get it and be fine, I suspect most of those will then decide to take it.”
 Ingham County, Mich., which includes Michigan State University, went from reporting about 300 new infections in August to about 1,800 in September.Credit…Sylvia Jarrus for The New York Times
Ingham County, Mich., which includes Michigan State University, went from reporting about 300 new infections in August to about 1,800 in September.Credit…Sylvia Jarrus for The New York TimesAs coronavirus deaths soar across the country, deaths in communities that are home to colleges have risen faster than the rest of the nation, a New York Times analysis of 203 counties where students compose at least 10 percent of the population has found.
In late August and early September, as college students returned to campus and some institutions put into place rigorous testing programs, the number of reported infections surged. Yet because serious illness and death are rare among young coronavirus patients, it was unclear at the time whether the growth of infections on campus would translate into a major health crisis.
But since the end of August, deaths from the virus have doubled in counties with a large college population, compared with a 58 percent increase in the rest of the nation.
Few of the victims were college students but, rather, older people and others living and working in the community.
Since the pandemic began, a Times survey has identified more than 397,000 infections at more than 1,800 colleges and universities. Those cases include more than 90 deaths involving college employees and students.
The link between an outbreak at a college and a coronavirus death in the wider community is often indirect and difficult to document, according to public health experts, especially without extensive contact tracing, which many local health departments in the United States lack resources to pursue.
But researchers have begun finding evidence of ties to college students. Using genetic sequencing to track cases around the University of Wisconsin-La Crosse, Paraic Kenny, a cancer geneticist at the Kabara Cancer Research Institute of the Gundersen Medical Foundation, has found links between infections at the university and cases and deaths in the surrounding region.
That trend highlighted a central fear of health officials — that young adults with limited symptoms might unwittingly transmit the virus, increasing the possibility it would ultimately spread to someone more vulnerable.
Experts suggest an array of ways such spread might happen, including one simple possibility: More than 1.1 million undergraduates work in health-related occupations, census data shows, including more than 700,000 that serve as nurses, medical assistants and health care aides in their communities.
But spread of the virus may also be more invisible, through layers of separation.
“All it really takes is one cavalier interaction,” said Tali Elfassy, an epidemiologist at the University of Miami.
GLOBAL ROUNDUP
 Members of the Public Health Agency of Canada held a drill for the rollout of the coronavirus vaccine in Ottawa on Friday.Credit…Pool photo by Sean Kilpatrick
Members of the Public Health Agency of Canada held a drill for the rollout of the coronavirus vaccine in Ottawa on Friday.Credit…Pool photo by Sean KilpatrickWith the F.D.A.’s authorization of a coronavirus vaccine on Friday, the United States became the third highly developed Western country — after Britain and Canada — to approve such a drug.
But the three countries have different health care systems and face different challenges in the race to get the vaccine to millions of people. Here are some similarities and differences:
For now, yes.
The first vaccine authorized by American regulators, and their British and Canadian counterparts, is the one developed by Pfizer and BioNTech.
But several other vaccines are close behind, particularly one developed by Moderna and the National Institutes of Health, and another from AstraZeneca and the University of Oxford. It could well be that half a dozen or more vaccines are approved in the coming months.
In Britain, very. In the United States, not. In Canada, somewhere in between.
With a strong central government and a National Health Service that covers all of its people, Britain is directing the process from London. The national government chose 50 hospitals that would initially get the vaccine and made sure they were prepared; decided how much each one would receive; and drafted rules determining what order people would be eligible to get it.
In the U.S., the federal government is having the vaccine distributed to each state based on population. It will be up to states to decide how to divide the doses among hospitals, clinics and, ultimately, drugstores and doctors’ offices.
Canada has a universal health care system, but it is decentralized, administered by provinces and territories. For vaccine distribution, the central government plans to work through those regional governments. Ottawa will play a large role in directing the process.
That remains a bit murky.
Canada ordered enough of the Pfizer-BioNTech vaccine for all of its people, Britain enough for 30 percent and the United States enough for 15 percent. But those numbers reflect deliveries that are expected to take months to complete, and Pfizer, like other companies, has hit snags in ramping up production.
All three countries made advance purchases from other companies, as well, so the pace of vaccine approval could significantly affect the speed of rollout. That speed will also be affected by the need for vaccination sites to be equipped with the right freezers, staff, and enough syringes and protective gear.
Initial shipments are a fraction of the pre-purchases — 800,000 doses to Britain and an expected 249,000 this month to Canada. U.S. officials said they hoped to have 40 million doses of the vaccine by the end of the month, which may be optimistic.
In Canada, the government is sending shipments to all 10 provinces. The three northern territories will have to wait. In America, FedEx and UPS will ship vaccines from distribution centers to every part of the country.
That is even murkier.
Britain, Canada and the United States have followed similar strategies, pre-ordering huge numbers of doses — more than enough to inoculate everyone — from multiple makers, hedging their bets in case some of the vaccines are not approved or some manufacturers have production breakdowns.
Relative to their populations, the United States has ordered far fewer doses than Canada or Britain, and last summer it passed up an offer to increase its advance order from Pfizer. Administration officials say the numbers are misleading, because the government has signed options to buy far more of the vaccine if it sees the need.
But in the face of intense global demand, it is not clear how fast pharmaceutical companies will be able to fulfill the orders they have, much less any additional orders.
In other global developments:
In Brazil, the number of people unwilling to take any Covid-19 vaccine has grown to 22 percent, up from 9 percent in August, Reuters reports. President Jair Bolsonaro, a virus skeptic who has continued to critique coronavirus restrictions, said he would not take a coronavirus vaccine. He has been particularly skeptical of China’s Sinovac vaccine. In November, Brazil suspended a late-stage trial of the vaccine. Although Brazil’s cases fell during the fall, there’s been an uptick since early November. It’s now averaging about 43,000 cases a day, about double from a month ago.
Brittanya Karma, a Vietnamese-German rapper and reality television star, died on Nov. 29 in Hamburg, Germany, where she was born and lived. She was 29. The cause was complications of Covid-19, her agent said.
Rizieq Shihab, a radical Indonesian cleric who held large gatherings and called for a “moral revolution” after his return from Saudi Arabia last month, surrendered to the police on Saturday on charges of violating coronavirus protocols. Upon his return, Mr. Rizieq, 55, invited 10,000 people to his daughter’s wedding. Authorities said six of his bodyguards were killed by the police in self-defense. In surrendering, he said he would cooperate with the investigation.
U.S. Roundup
 The Illinois National Guard, at a testing center in May, are assisting with coronavirus testing and screening at the LaSalle Veterans’ Home. At least 38 residents have tested positive for the virus.Credit…Nam Y. Huh/Associated Press
The Illinois National Guard, at a testing center in May, are assisting with coronavirus testing and screening at the LaSalle Veterans’ Home. At least 38 residents have tested positive for the virus.Credit…Nam Y. Huh/Associated PressA coronavirus outbreak at a veterans’ home in Illinois has killed more than a quarter of its residents, prompting the state to mobilize the Illinois National Guard to the home, Gov. J.B. Pritzker said at a news conference on Thursday.
Mr. Pritzker said that 38 residents at the home, LaSalle Veterans’ Home, had contracted the virus out of its 93 total residents, and three staff members were also infected. The governor emphasized that every employee was tested with rapid result antigen tests before each shift, and that residents were being tested every day.
Still, some are criticizing the handling of the outbreak, which was first detected at the home on Nov. 1. Since then, 89 percent of the residents have tested positive for the virus, State Senator Sue Rezin, a Republican, said in a letter on Friday addressed to the veterans at the home. Ms. Rezin said that National Guard medical staff would be assisting with testing and screening, and she noted that the move to mobilize the National Guard was made five weeks after the initial outbreak.
“The fact that this virus was able to run rampant in the facility should be considered unacceptable,” Ms. Rezin wrote in the letter. “With so many unanswered questions remaining, it is critical that the legislature continue its legislative hearings into what happened at the LaSalle Veterans’ Home,” she said.
There are continuing investigations into the outbreak. On Monday, the governor’s office fired the administrator at the LaSalle home and the director of nursing had also been placed on administrative leave. The Illinois Department of Veterans’ Affairs released reports in November, in partnership with the state department of public health and the U.S. Department of Veterans Affairs, detailing several problems in the home.
Alcohol-free sanitizer — which is not effective in eradicating the virus — was stocked in mounted dispensers, a report said. Some staff members were seen in the kitchen area with masks pulled down to their chins and not social distancing. There were staff members wearing gloves, who were seen touching patients and multiple surfaces “without changing or performing hand hygiene,” the report said. “Opportunities for transmission among some staff may have occurred due to reported laxity of masking and social distancing while off duty and also during break periods.”
Illinois has seen a 10 percent decline in cases over the past 14 days, and a 14 percent decline in hospitalizations, after setting seven-day case records at the beginning of November. Yet deaths in the state are up 42 percent in the past two weeks.
The F.D.A. authorized Pfizer’s vaccine for emergency use on Friday, allowing for millions of people to start getting the vaccine within days. Still, the virus continues to decimate the lives of residents at the LaSalle Veterans’ Home, even as residents of nursing homes and long-term-care facilities — and health care workers at high risk of exposure — are at the top of the vaccine waiting list.
In other news around the United States:
Dr. Nicole Alexander-Scott, the director of the Rhode Island’s Department of Health, tested positive for the coronavirus on Saturday. The results came from routine testing, a spokesman for Gov. Gina Raimondo said. Dr. Alexander-Scott was asymptomatic. Ms. Raimondo, who last saw Dr. Alexander-Scott in person during a Covid-19 press briefing on Thursday, is quarantining. She tested negative on Saturday, spokesman Josh Block said. Rhode Island leads the country in coronavirus cases per capita, averaging about 1,240 cases a day this past week, or about 117 cases per 100,000 people. The state has begun issuing temporary licenses to doctors and nurses who are retired, are visiting the state or have recently completed training programs.
As many as 300,000 coronavirus cases across the United States can be traced to a two-day conference in Boston attended by 175 biotech executives in February, according to a study published in the journal Science.
 A vaccination station in Cardiff, England, where the Pfizer and BioNTech vaccine was given to patients this week.Credit…Andrew Testa for The New York Times
A vaccination station in Cardiff, England, where the Pfizer and BioNTech vaccine was given to patients this week.Credit…Andrew Testa for The New York TimesBritish drug regulators recommended on Wednesday that people with severe allergic reactions to food and medicine not receive Pfizer’s coronavirus vaccine after two health care workers, both with such a history, had a serious reaction.
The initial report on the British cases set off alarm and confusion, and left many people with allergies wondering whether the new vaccine would be safe for them.
Here’s what we know so far:
The workers both experienced anaphylaxis, a life-threatening reaction that impairs breathing and can drop blood pressure within minutes or even seconds after exposure to an allergen. Both workers were treated with epinephrine and have recovered, the regulators said.
People with a history of an anaphylactic reaction to any vaccine were excluded from Pfizer’s studies, company officials said on Thursday.
Among those who participated in the Pfizer trials, a very small number of people had allergic reactions. A document published by the F.D.A. on Tuesday said that 0.63 percent of participants who received the vaccine reported potential allergic reactions, compared with 0.51 percent of people who received a placebo.
In Pfizer’s late-stage clinical trial, one of the 18,801 participants who received the vaccine had an anaphylactic reaction, according to safety data published by the F.D.A. None in the placebo group did.
I have allergies, should I be concerned?
Dr. Anthony Fauci, the nation’s leading expert on infectious diseases, said on Wednesday that the allergic reactions were concerning but most likely rare, the kind of effects that show up when a vaccine moves out of testing and into broader distribution.
“If I were a person that had an underlying allergic tendency, I might want to be prepared that I might get a reaction, and therefore be ready to treat it,” Dr. Fauci said, in a webcast moderated by Dr. Sanjay Gupta of CNN, sponsored by Harvard and The New England Journal of Medicine.
Dr. Fauci acknowledged that the problem could turn out to affect a lot of people, but he said that other vaccines would eventually become available for those affected.
Dr. Paul Offit, a vaccine expert at Children’s Hospital of Philadelphia, said the initial, broad recommendation in Britain mentioning severe allergic reactions seemed to be an overreaction that could needlessly scare many people away from a desperately needed vaccine in the middle of a raging pandemic.
Fewer than one in a million recipients of other vaccines a year in the United States have an anaphylactic reaction, Dr. Offit said. Those reactions are treatable and much easier to control than a severe case of Covid-19, he said.
 The stolen ventilators, worth $3 million, were being transported to a Covid-19 intensive care center in El Salvador to treat critically ill patients as part of a federal aid program.Credit…Go Nakamura/Getty Images
The stolen ventilators, worth $3 million, were being transported to a Covid-19 intensive care center in El Salvador to treat critically ill patients as part of a federal aid program.Credit…Go Nakamura/Getty ImagesTwo men have been charged with stealing 192 ventilators owned by the U.S. government in Miami, authorities said on Friday. When they were stolen, the ventilators — worth $3 million — were being transported to a Covid-19 intensive care center in El Salvador to treat critically ill patients as part of a federal aid program.
Yoelvis Denis Hernandez, who is known as Guajiro, and Luis Urra Montero, who goes by the name Flaco, were indicted on charges of federal conspiracy, possessing stolen goods that were being shipped across state lines and the theft of government property.
According to the indictment, Mr. Hernandez, 42, and Mr. Montero, 24, stole a tractor-trailer in August that was being used to transport the ventilators to Miami International Airport. The tractor-trailer had been left in a lot overnight. After an investigation, most of the ventilators were found, the authorities said.
At the onset of the pandemic, medical professionals and government authorities were concerned about the shortage of ventilators in the United States and in other countries. Since then, however, the country’s medical device manufacturers have ramped up production of the machines. The U.S. now has more than 200,000 critical care ventilators — 155,000 of them in the Strategic National Stockpile — but there aren’t enough specialists to operate them.
In June, a man was charged with stealing ventilators from a Veterans Affairs medical center in Seattle and selling them on eBay.
as per our monitoring this Story originally appeared * : ) here → *
Covid-19 Updates: First U.S. Vaccines Are About to Be Shipped as Virus Ravages America
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

Teva to be Sole Distributor of COVID-19 Vaccines in Israel
Teva to be Sole Distributor of COVID-19 Vaccines in Israel
Photo Credit: Yonatan Sindel / Flash 90

 TEVA Pharmaceutical Industries headquarters in Jerusalem
TEVA Pharmaceutical Industries headquarters in Jerusalem The Israeli Teva Pharmaceuticals firm has signed an exclusive agreement with the government to distribute all the COVID-19 vaccinations in the Jewish State.
Teva Israel CEO Yossi Ofek told the Globes business news site on Sunday, “We are waiting for the green light and the possibility that within several weeks millions of vaccination shots will arrive here.
Advertisement <!–Publisher #16: JewishPress.com Zone #113: Comment Banner / (02) / News Size #15: Banner 468×60 (Comments and Mobile) [468×60] –>
“It’s looking like Israel will be the first place after the US that will receive vaccinations for a large part of the population, even before Britain.
“But since the announcement the vaccine shots are set to arrive in larger numbers than expected. We have strengthened the readiness at TEVA SLA’s logistics center in Shoham and Teva’s center in Kfar Saba.
“On both these sites there are already freezers that can chill Pfizer’s vaccinations to minus 70 degrees centigrade, and we are in the process of obtaining more freezers, so that within several weeks we can take in three million vaccination doses at this temperature. Moderna’s vaccination is kept at a less low temperature and we are already prepared to receive them today,” he said.
“This is something that we have not ever previously experienced in this field,” Ofek underlined. “We have developed methods in the country to cope with it, from the moment that the plane leaves the factory to us. The process is very complicated and has to be 100 percent successful.”
The plan entails distributing the vaccine to hundreds of clinics, care homes and possibly other institutions as well. At these destinations, the HMO takes responsibility for the package, but from the moment the mini-package comes out of the freezer, it can be kept at the typical temperature for vaccinations of 2 to 8 degrees centigrade for up to five days only. In each package there will be 970 doses, meaning every package will hold enough for 970 Israelis to be vaccinated.
The Health Ministry is expected to give the green light to vaccinate Israel’s population as soon as the FDA approves the vaccine, Ofek said.
“We have responsibility for any defect in the product during the period it is in our hands,” he told Globes. “Regarding responsibility for the product itself, for the efficacy and safety of it, these are part of agreements between Israel and Pfizer and Moderna, in which we are not involved.”
Advertisementas per our monitoring this Story originally appeared * : ) here → *
Teva to be Sole Distributor of COVID-19 Vaccines in Israel
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic

Coronavirus updates: California region has 0.0% available ICU capacity; US reports 16 millionth case; vaccinations to begin Monday
Coronavirus updates: California region has 0.0% available ICU capacity; US reports 16 millionth case; vaccinations to begin Monday
CLOSE![]()
The COVID-19 vaccine is using new technology that has never been used before in traditional vaccines. Here’s how an mRNA vaccine works. USA TODAY
The United States on Saturday reported its 16 millionth COVID-19 case, even as a newly authorized vaccine is already being distributed and vaccinations are expected to begin Monday, according to federal officials.
It took three months for the U.S. to record its first million cases. But it’s been just four days between the 15th and 16th million case milestones — yet another sign the virus is spreading at an alarming and deadly pace. But the nation’s first authorized vaccine promises to help slow the pandemic in coming months, as the federal government races to quickly distribute it.
“As I speak today, right now, vaccines are being packaged,” Gen. Gustave Perna, co-leader of Operation Warp Speed in charge of logistics, said Saturday. “Tomorrow morning, vaccines will start rolling from manufacturing to distribution hubs. By Monday, vaccines will be received.”
Vaccinations will begin Monday, Dr. Nancy Messonnier, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during a meeting Saturday.
Health care workers and nursing home residents are first in line. Patients and caregivers will receive fact sheets describing the risks and benefits, she said.
Earlier Saturday, Food and Drug Administration officials reassured the American public that the agency did not cut corners in its review and urged people to get vaccinated. The United States is on the cusp of losing 300,000 people to COVID-19.
Keep refreshing this page for the latest updates. For headlines in your inbox, sign up for our newsletter, Coronavirus Watch.
Here are today’s top headlines:
- An advisory committee to the Centers for Disease Control and Prevention voted Saturday to recommend the Pfizer/BioNTech vaccine for people 16 and older. Now it goes to the director of the CDC for his signature.
- The Food and Drug Administration late Friday granted emergency authorization to Pfizer/BioNTech’s vaccine that research shows is highly effective at preventing a disease.
- The promising news will not immediately end the pandemic, which is raging out of control. Dr. Ugur Sahin, CEO of BioNTech, estimated it would be March or April before the vaccine could impact the virus’ spread — underscoring the need for measures like mask-wearing and social distancing in the meantime.
- About 1 in 8 U.S. hospitals had few or no intensive care unit beds available last week, according to new federal data. Experts say the number of hospitals struggling to accommodate the nation’s sickest patients likely will increase following another week of record COVID-19 cases.
- Two Florida men are facing federal charges that allege they stole nearly 200 ventilators the United States shipped to El Salvador to help the Central American country treat coronavirus patients.
Another day of record deaths in the US: On Friday, 3,309 people in the U.S. died from the coronavirus, according to Johns Hopkins University data. The previous record was set Wednesday at 3,124, the first time the daily death toll surpassed 3,000. As of Saturday, about 297,000 people in the U.S. have died, with more than 16 million confirmed cases. The global totals: 71.6 million cases and 1.6 million deaths.
What we’re reading: We’re answering your questions about the vaccine, like: What are the side effects? Can you still get sick? Is it safe during pregnancy? Read more here.
New York’s 21 Club closes indefinitely due to pandemic
The storied 21 Club in midtown Manhattan, a favorite of celebrities and the power elite for nine decades, is closing indefinitely due to the coronavirus pandemic, but the owners are optimistic about reopening at some point.
The restaurant’s owners filed notice about the closing with the city on Wednesday, saying all 148 employees will be terminated on March 9. The news comes as indoor dining at New York City restaurants will be banned again starting Monday in an effort to halt a resurgence of the virus, but takeout and outdoor dining will still be allowed.
The restaurant has been closed since last March during the first wave of the pandemic, as indoor dining was prohibited.
The 21 Club was a favorite dining place of presidents since Franklin D. Roosevelt’s day. Shortly after the 2016 election, then-President-elect Donald Trump broke protocol by leaving his Trump Tower residence without taking along his press contingent to eat at the restaurant. John Steinbeck, Ernest Hemingway and Frank Sinatra had favorite tables at 21, and Humphrey Bogart proposed to Lauren Bacall there. It appeared in films ranging from the 1950 Bette Davis classic “All About Eve” to 1987’s “Wall Street.”
— Associated Press
12-county California region has 0.0% available ICU capacity
In San Joaquin County, part of California’s vast Central Valley that produces most of the country’s fruits and vegetables, the coronavirus is spreading like a weed and the hospitals are running out of beds for the sickest patients.
San Joaquin is part of a 12-county region that on Saturday, according to the California Department of Public Health, had 100% of its intensive care unit beds filled, the highest rate anywhere in California. And with cases continuing at an unprecedented rate, the death toll inevitably will grow, too.
A new stay-at-home order was imposed this week but it is unknown whether it will have the intended consequence of finally changing enough people’s behavior to slow infections as a vaccine is widely rolled out.
— The Associated Press
UPS Worldport hub in Louisville to soon ship vaccine
The first shipments of Pfizer/BioNTech’s COVID-19 vaccine will move through UPS’ Worldport global air hub at the Louisville, Kentucky, airport Sunday, a company spokesman told The Courier Journal, part of the USA TODAY Network.
UPS said the vaccine will originate from storage sites in Michigan and Wisconsin.
“The vaccines will be transported to UPS Worldport facilities in Louisville, where they will be expedited Next Day Air to select destinations, including hospitals, clinics and other medical facilities, to inoculate healthcare workers,” the company said in a news release.
The shipments will arrive at the hub to be sorted and will be shipped out — all on Sunday, UPS spokesman Jim Mayer said. He said he did not know how many doses will be moving through. Mayer previously said that UPS will deliver to states in the eastern half of the United States. FedEx will deliver to the western half of the U.S.
– Ben Tobin, Louisville Courier Journal
‘We are not taking a victory lap,’ Operation Warp Speed official says
The chief operating officer of Operation Warp Speed, the White House effort to fund, produce and distribute a vaccine, cheered the emergency authorization of the Pfizer/BioNTech vaccine but warned of a tough road ahead.
“While this moment is extraordinary … we have a lot of work to do. We are not taking a victory lap,” Perna said Saturday.
He said it wouldn’t have made a difference to Operation Warp Speed if the FDA had authorized the vaccine Saturday instead of Friday. “Our ultimate goal was to get it there no later than Monday morning,” he said.
Perna reiterated that Operation Warp Speed wouldn’t distribute the second vaccine dose or a portion held in reserve until it has “ultimate confidence” that there are enough doses and the delicate distribution system works. That could change in January or February.
“We want no vaccines on a shelf,” Perna said. “Doesn’t matter how effective a vaccine is if it’s on a shelf.”
Perna said vaccine doses were not positioned in advance because the operation “did not want to presume” emergency use authorization. “Under no circumstances did we want to get ahead of the great FDA and their decision making,” he said.
FDA: No corners cut in reviewing Pfizer/BioNTech vaccine
Amid concerns about how quickly the FDA issued an emergency authorization for the Pfizer/BioNTech vaccine, agency leaders stressed Saturday that they conducted a thorough, transparent review.
“Science and data guided the FDA’s decision. We worked quickly because of the urgency of this pandemic, not because of any other external pressure,” FDA Commissioner Stephen Hahn said. “I will absolutely take this COVID-19 vaccine.”
Hahn said reports Friday that the White House had threatened to fire him if the agency did not authorize the vaccine were “inaccurate.”
Hahn said the agency was “very concerned about vaccine hesitancy” and made the process as transparent as possible by posting trial data and documents online.
“Efficiency does not mean any cutting of corners,” Hahn said. The FDA “found ways to cut the red tape,” but “important safety checks remained in place,” he said.
Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said the FDA requires vaccine developers to monitor for “any significant adverse events” as they seek standard approval. The FDA and the CDC are monitoring as well, he said.
Officials provided more information about whether certain groups of people should get the vaccine. People who are pregnant or immunocompromised, who were not included in safety trials, should discuss the vaccine with their providers “on an individual basis,” Marks said.
Vaccination sites will be equipped to treat allergic reactions, official says
Sites where the Pfizer/BioNTech vaccine are administered will be able to treat allergic reactions, Marks said. About 1.6% of the population has had a severe allergic reaction to food or something in the environment but are eligible to receive the vaccine, Marks said. Only people who have had a severe allergic reaction to a previous dose of the vaccine or one of its components should not receive the vaccine.
Experts say the ingredients in the Pfizer/BioNTech vaccine look typical: an active ingredient (in this case, messenger RNA), fats, salt and sugars.
Dr. Matthew Heinz, a hospitalist based in Tucson, Arizona, said if any part of the vaccine were to trigger an allergic reaction, it could be one of the components of the fat molecule. But that’s rare. “We’re talking about a number of relatively mild reactions that you can count on one hand,” out of tens of thousands of people in the study, he said.
Two British people with severe allergies apparently had reactions to Pfizer/BioNTech’s vaccine this week. Allergic reactions were not a significant problem in the U.S. trial, in which more than 20,000 people received two doses of the vaccine. The U.S. trials kept out subjects who have had severe allergic reactions.
— Adrianna Rodriguez
Friday set another record for COVID-19 cases, deaths
On the same day that the FDA approved the first vaccine for emergency use in the United States, the nation reached another milestone in COVID-19 cases and deaths.
The country recorded 3,309 coronavirus deaths Friday, according to Johns Hopkins University data, surpassing Wednesday’s record when 3,124 people died. Friday also saw the highest number of daily cases to date in the U.S.: 231,775.
Pfizer’s COVID-19 vaccine gets FDA authorization
In what is hoped to be the beginning of the end of America’s COVID-19 pandemic, the FDA on Friday evening authorized the first vaccine to prevent people from getting sick.
What this means: The Pfizer/BioNTech vaccine received emergency use authorization, which is not full approval. Although it has received all the standard short-term safety and effectiveness reviews, the vaccine has not been tested for the two years typical of an approved vaccine. So it is not yet clear how long protection will last.
When will you get it? Frontline health care workers and nursing home residents are expected to get the vaccine first. More doses will be rolled out in the weeks and months to come, with Pfizer and Moderna each expected to deliver 100 million doses of their vaccines by the middle of next year.
What about other vaccines? Next week, a similar COVID-19 vaccine developed by the Massachusetts-based Moderna will go through the same review process, and could swiftly be cleared for use.
– Karen Weintraub
In America’s hardest-hit county, COVID deaths claimed parents, friends
Just over a month ago, the coronavirus pandemic tore through Quinter, Kansas, a rural town of 1,000, killing 20 residents. Here, where most everyone knows most everyone else, the pandemic has killed farmers and their wives. The town’s unofficial historian. The beloved grandmother whose sour cream chocolate cake with chocolate fudge frosting was always the talk of the party. The mom whose piano-playing still echoes in the heads of her friends.
And it has drained the hearts of the survivors. Those who feel guilty about recovering. The ambulance workers battling to treat their own relatives. The exhausted doctor who watched nearly half his patients die.
As of Thursday, the coronavirus has killed a higher percentage of Gove County residents than any other county in the United States: one out of every 132 people. Even today, mask-wearing remains controversial. Friendships are strained as authorities struggle to persuade their neighbors to follow basic public health guidelines, such as avoiding large gatherings.
“We are living through history right now, and I worry what the history books will say about us,” said Ericka Nicholson, 47, who helps run the town’s volunteer ambulance service and survived the infection. Read more from Gove County, Kansas.
– Trevor Hughes
CLOSEWe asked you to tell us your biggest questions about the COVID-19 vaccines. Here are some answers. USA TODAY
Relentless coronavirus surge fills 1 in 8 hospital ICU units
About 1 in 8 U.S. hospitals had little or no intensive care unit space available last week. Experts say the number of hospitals struggling to accommodate the nation’s sickest patients likely will increase after another week of record COVID-19 cases.
The federal government this week released a sweeping database showing a one-week average of COVID-19 patients in hospitals nationwide. It’s the first time the Department of Health and Human Services has provided such detailed information on nearly 5,000 U.S. hospitals since the pandemic began.
The absence of hospital-level information has been a blind spot in the pandemic, as administrators must call neighboring hospitals to transfer patients when they run out of beds and staff.
“We know what’s happening in our system, but I don’t know what’s happening in the other systems,” said Dr. Lewis Kaplan, professor of surgery at University of Pennsylvania Hospital in Philadelphia. “To have a coordinated effort you need to have that kind of data so everyone knows where everyone else happens to be.” Read more.
– Ken Alltucker and Aleszu Bajak
US buys another 100M doses of Moderna’s vaccine
The Trump administration announced Friday that it has purchased an additional 100 million doses of a coronavirus vaccine made by Moderna, bringing the federal government’s total order from the company to 200 million doses. The additional doses would “provide for continuous delivery through the end of June 2021,” the Department of Health and Human Services said in a press release.
“Securing another 100 million doses from Moderna by June 2021 further expands our supply of doses across the Operation Warp Speed portfolio of vaccines,” HHS Secretary Alex Azar said in a statement. “This new federal purchase can give Americans even greater confidence we will have enough supply to vaccinate all Americans who want it by the second quarter of 2021.”
Moderna became the second vaccine maker in the U.S. to request authorization from the FDA at the end of last month. The company’s latest findings showed that of 196 people in the clinical trial who caught COVID-19, 185 of them had received the placebo, while only 11 had received the vaccine. That works out to an effectiveness rate above 94%.
Wisconsin woman reunites with family after battling virus for nearly 3 months
After spending over 80 days battling COVID-19 in a Wisconsin hospital, Nancy Van Dyn Hoven on Friday was reunited with her family and friends — just in time for the holidays.
“Other than 2020 being awful, this is just the best,” Van Dyn Hoven, 60, said Friday at home, chuckling with her husband, Dennis, and daughter, Stacy Arnoldussen. “It’s all I could ask for.”
Dr. Anthony Zeimet, an infectious diseases specialist at the hospital and Nancy’s doctor, said the severity of her illness was particularly surprising because she was just 59 years old — she turned 60 in the hospital — and had no underlying health conditions that would put her at higher risk of severe illness.
“The virus kind of ravaged her body,” Zeimet said. “It just goes to show that, with COVID, we don’t know who’s going to do well or do poorly. Nancy was someone who, when she was first admitted, we thought she’d do pretty well. … Unfortunately, she ended up being here for 80-plus days.”
Although Nancy’s recovery is far from over and she has a long road of rehabilitation services ahead, Friday marked the end of a months-long struggle with the virus. .
— Samantha West, Appleton Post-Crescent






















































Artists Jack Schwab, and Debbie Wilger, wear their masks July 14, 2020, inside the Missouri Artists on Main store in downtown St. Charles, Mo. Schwab, 60, who makes silver jewelry, and Wilger, 63, a painter, are concerned about the uptick in coronavirus cases in St. Charles County, and say most customers in the store abide by their facial covering policy, but a few have left in anger because of it. Jim Salter, AP
































Alice Mayes, 92, is visited by her family at Signature HealthCARE on May 6, 2020 in NewBurgh, Ind. The family, from left, Onya Rhoades, Lexi Rhoads, 3, Dylan Rhoades, 5, Kaitlyn Helmbrecht, 2, James Helmbrecht and Del Mayes were separated by a window glass on May 6, 2020 in Newburgh, Ind. The 92-year-old is a COVID-19 survivor. Denny Simmons, Evansville Courier & Press

































Austin High School seniors and best friends, clockwise from top left, Brooke Peterman, 17, Maddy McCutchin, 18, Lucia Saenz, 17, Reese Simek, 18, and Lily Tickle, 18, visit with each other in the parking lot at the school in Austin, Texas, on Sunday April 5, 2020. In the midst of a shelter in place order due to the coronavirus pandemic, the girls sat in the back of their cars to chat at a safe distance. Jay Janner, Austin American-Statesman / USA TODAY Network








Replay
Autoplay
Show Thumbnails
Show Captions
Read or Share this story: https://www.usatoday.com/story/news/health/2020/12/12/covid-vaccine-update-pfizer-vaccine-authorized-first-doses-coming/3893846001/
as per our monitoring this Story originally appeared * : ) here → *
Coronavirus updates: California region has 0.0% available ICU capacity; US reports 16 millionth case; vaccinations to begin Monday
SCUC-CCP :: Selective Cause Until it is Concluded : Coronavirus Covid-19 Pandemic
*Above All Stories ON/ABOUT/FROM ‘Coronavirus Covid-19 Pandemic’ are Published/Edited/Enhanced randomly by the Global Open Profile: allaboutian.

 scroll for the Story
scroll for the Story
~ MORE ~
explore a.
→ all Story at a.
✐ Publish
?️?️ Perceive
? Play
TrendingStory
SocialStory
RandomStory

More SCUC
Suggested SCUC
Suggest SCUC
*If You think there is a certain Cause in this planet which should be concluded immediately. You may Suggest that here as ‘Suggested SCUC’.
*Select Category/Tag: Suggested SCUC , Put A Clear Title, Upload an Image & Give A brief/details Description to portray the Cause at your next publish screen.